  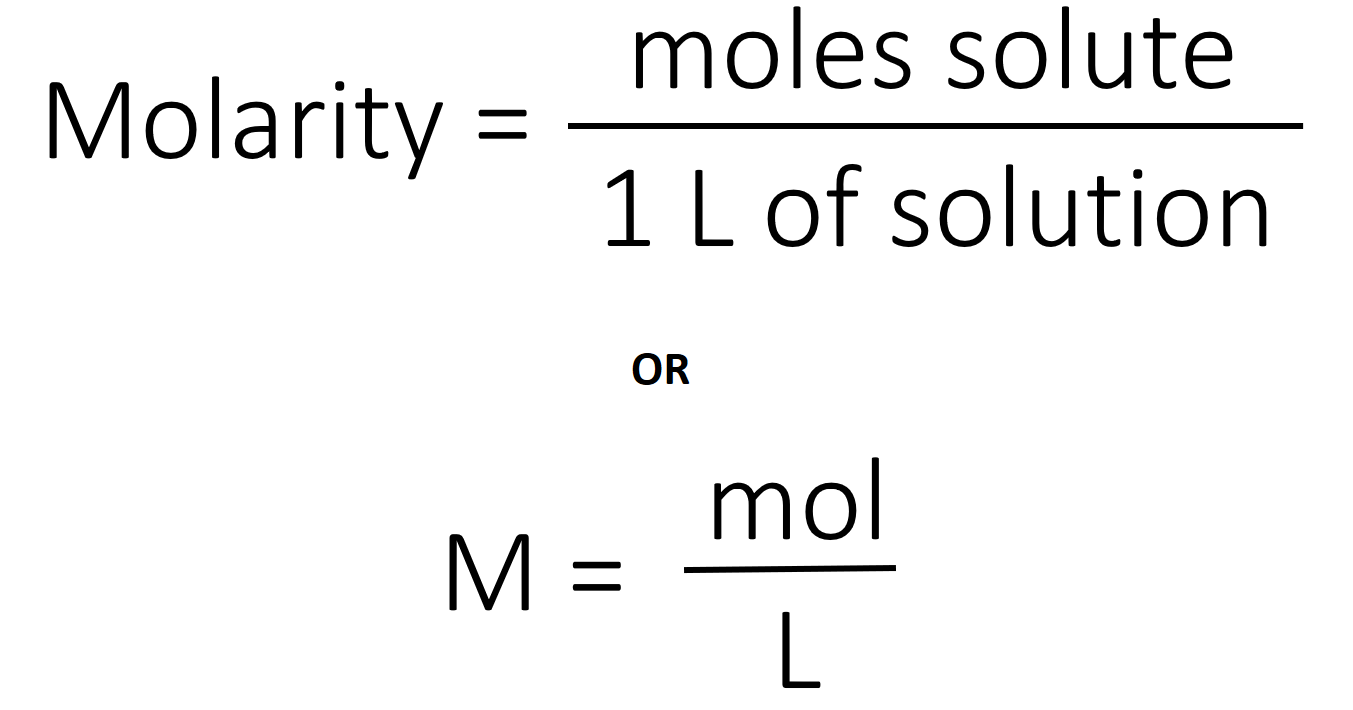
The easiest way to understand the process is to assume you have 1L of solution which is 1000 mL. So, in this way we can find the mass of a mole fraction.If all you are given is Molarity, then you also need to know the density of the solution to calculate molality, m (mol/kg solvent). On addition of all the masses we get 1160g The total of mole fraction comes up to be 1.0. We can understand the interconversion of the two quantities from the following example:įind the mole fraction: moles / total moles Read more about: Is Hydrogen Bond Stronger Than Covalent: Why, How and Detailed Facts How to find the mass of a mole fraction? Therefore Molarity = XB × 1000 / ( 1- XB ) × MAīy rearranging the above equation in an order of our requirements we can calculate mole fraction. XB × 1000 / XA × MA = WB × 1000 / WA × MB = m = Molarity Let us now see what is the relation between mole fraction and molarity.Īfter dividing equation 1 by equation 2 we get Read more about: How To Find Molar Mass From Volume: Detailed Explanations How to find mole fraction from molarity? Find out number of moles in 35g of glucose Find out the number of moles in 70g of CH4.įirst, we should calculate molar mass ( by simply adding the weight of the atoms in CH4 formula) and we are provided with mass = 70g (m)Ģ.We can find moles from the molar mass by using the formula given below:įor better understanding let’s see some examples: We know that 1 mole is equivalent to 6.022 × 10 23. So, if we know density and molar mass of a substance we can easily calculate its molarity. So now we know the perfect meaning of the three terms. The standard unit used to describe molar mass is kg/mol. It’s sometimes also referred to as molecular weight. The Molar mass of a compound ( chemical) is the mass of the compound ( in consideration) divided by the substance ( amount) in the sample of the compound being referred. Temperature is inversely proportional to density meaning as the temperature is increased ( keeping the pressure constant) the density will decrease. So in this way by using the above relationship we can find out molar mass from molarity. 
The provided Volume make sure before substituting it should be in liters.Īfter knowing the three quantities, we can substitute them in equation 2 and obtain molar mass. Molarity = moles of solute / Liters of solution or Volume of solution.īy using the above relation we can calculate molarity. There are many ways in which we can find or calculate molarity but the very basic one that we use nowadays is: So we can find the three quantities the weight of the solute ( w ), Molarity ( M ), and Volume of the solution ( V ). We can find molar mass from molarity by rearranging the equation 1 
V = volume of the final solution once the complete dissolution is done ( in litres ). W = the weight of the solute ( in grams ) To understand how to find molar mass from molarity we must first understand the relationship between the two terms. For this we should know the other quantities other than the one of our interest. There are many ways to find molar mass from molarity, but we are going to work with one equation from which on rearranging it we can obtain the required product. In this article we are going to analyze how to find molar mass from molarity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
